Furagin: Forced Degradation Studies
Furagin: Forced Degradation Surveies
Index
Index of Figures
Index of Tables
Abstraction
Keywords
Abbreviations
1.Introduction
1.1.Forced debasement surveies
1.1.1. Overview of regulative counsel
1.1.2. Review of bing literature and different attacks
1.2.Furagin
1.2.1. Chemical and Physical Information
1.2.2. Pharmaceutical Information
1.2.3. Impurities
2.Aim of the Study
3.Method
3.1.Equipment and stuffs
3.1.1 Method AHPLC/AUPLC
3.1.2 Method B
3.2.Reagents
3.2.1 Method AHPLC/AUPLC
3.2.2 Method B
3.3.Working criterions
3.3.1 Method AHPLC/AUPLC
3.3.2 Method B
3.4.Preparation of the solutions
3.4.1 Method AHPLC/AUPLC
3.4.2 Method B
3.5.Chromatographic conditions
3.5.1 HPLC conditions – Method AHPLC
3.5.2 UPLC conditions – Method AUPLC
3.5.3 UPLC conditions – Method B
3.6.Injection Sequence and System Suitability
3.7.Calculation and look of the consequences
3.7.1 Method standardization
3.7.2 Evaluation of extremum pureness
4.Forced debasement survey
4.1.Acid Hydrolysis ( AH )
4.2.Base Hydrolysis ( BH )
4.3.Oxidation – H2O2
4.4.Oxidation – AIBN
4.5.Thermal Hydrolysis ( solution )
4.6.Thermal ( solid )
4.7.Thermal/Humidity ( solid )
4.8.Photolytic
4.8.1 Solid
4.8.2 Solution
5.Results
5.1.Acid Hydrolysis – Room Temperature
5.2.Acid Hydrolysis – 50°C
5.3.Base Hydrolysis
5.4.Oxidation – H2O2
5.5.Oxidation – AIBN
5.6.Thermal Hydrolysis ( solution )
5.7.Thermal ( solid )
5.8.Thermal/Humidity ( solid )
5.9.Photolytic – Solid
5.10.Photolytic – Solution
6.Discussion
Mentions
7.Acknowledgments
Appendix I
Index of Figures
Figure 1: Flow diagram with the different forced debasement conditions to be used for drug substances and drug merchandises ( adapted from 16 )
Figure 2: Flow chart for executing emphasis surveies for hydrolytic debasement under acid and alkali conditions.
Figure 3: Flow chart for executing emphasis surveies for hydrolytic debasement under impersonal conditions.
Figure 4: Flow chart for executing emphasis surveies for debasement under oxidative conditions.
Figure 5: Flow chart for executing emphasis surveies for photolytic debasement.
Figure 6: Furagin
Figure 7: 1- { ( E ) – [ ( 2E,4E ) -5- ( 5-nitrofuran-2-yl ) penta-2,4-dien-1-ylidene ] amino } -imidazolidine-2,4-dione
Figure 8: Chemical reaction strategy of dross A
Figure 9: 1- [ ( Z ) -3- ( 5-Nitro-furanyl-2 ) -propene-2- ( Z ) -ylidenamino ] -imidazolidine-2,4-dione
Figure 10: Chemical reaction strategy of dross B
Figure 11: Chemical reaction strategy of the formation of Furagin from Impurity B
Figure 12: 5-Nitrofuran-2-carbaldehyde
Figure 13: Chemical reaction strategy of dross C
Figure 14: 3- ( 5-Nitrofuran-2-yl ) prop-2-enal
Figure 15: Chemical reaction strategy of dross D
Figure 16: Chemical reaction strategy of the formation of dross D by acid catalysed hydrolysis of Furagin
Figure 17: 1- { ( E ) – [ ( 5-nitrofuran-2-yl ) methylidene ] amino } -imidazolidine-2,4-dione
Figure 18: Chemical reaction strategy of dross E
Figure 19: { ( 2E ) -1-carbamoyl-2- [ ( 2E ) -3- ( 5-nitrofuran-2-yl ) prop-2-en-1-ylidene ] hydrazinyl } -acetic acid
Figure 20: Chemical reaction strategy of dross F
Figure 21: Chemical reaction strategy of the formation of dross F by basic catalysed hydrolysis of Furagin
Figure 22: 1-Aminoimidazolidine-2,4-dione hydrochloride
Index of Tables
Table 1: Potential Adverse Effects of Instability in Pharmaceutical Merchandises
Table 2: Conditionss Generally Employed for Forced Degradation
Table 3: HPLC analysis conditions used – Method AHPLC
Table 4: Gradient plan used in the HPLC analysis – Method AHPLC
Table 5: Approximately keeping clip, comparative keeping clip ( RRT ) and comparative response factor ( RRF ) of compounds – Method AHPLC
Table 6: UPLC analysis conditions used – Method AUPLC
Table 7: Gradient plan used in the UPLC analysis – Method AUPLC
Table 8: Approximately keeping clip, comparative keeping clip ( RRT ) and comparative response factor ( RRF ) of compounds – Method AUPLC
Table 9: UPLC analysis conditions used – Method B
Table 10: Approximately keeping clip, comparative keeping clip ( RRT ) and comparative response factor ( RRF ) of compounds – Method B
Table 11: Stress types and conditions for forced debasement survey of Furagin
Table 12: Consequences of acerb hydrolysis at room temperature of Furagin obtained by HPLC
Table 13: Consequences of acerb hydrolysis at room temperature of Furagin obtained by UPLC
Table 14: Consequences of acerb hydrolysis at 50°C of Furagin obtained by HPLC
Table 15: Consequences of acerb hydrolysis at 50°C of Furagin obtained by UPLC
Table 16: Consequences of basal hydrolysis of Furagin obtained by HPLC
Table 17: Consequences of basal hydrolysis of Furagin obtained by UPLC
Table 18: Consequences of oxidization with H2O2 of Furagin obtained by HPLC
Table 19: Consequences of oxidization with H2O2 of Furagin obtained by UPLC
Table 20: Consequences of oxidization with AIBN of Furagin obtained by HPLC
Table 21: Consequences of oxidization with AIBN of Furagin obtained by UPLC
Table 22: Consequences of thermic hydrolysis of Furagin solution obtained by HPLC
Table 23: Consequences of thermic hydrolysis of Furagin solution obtained by UPLC
Table 24: Consequences of thermic hydrolysis of Furagin pulverization obtained by HPLC
Table 25: Consequences of thermic hydrolysis of Furagin pulverization obtained by UPLC
Table 26: Consequences of thermic hydrolysis in the presence of humidness of Furagin pulverization obtained by HPLC
Table 27: Consequences of thermic hydrolysis in the presence of humidness of Furagin pulverization obtained by UPLC
Table 28: Consequences of photolytic debasement of Furagin pulverization obtained by HPLC
Table 29: Consequences of photolytic debasement of Furagin pulverization obtained by UPLC
Table 30: Consequences of photolytic debasement of Furagin solution obtained by HPLC
Table 31: Consequences of photolytic debasement of Furagin solution obtained by UPLC
Abstraction
T.
Aim: .
Consequences: .
Decisions: .
Keywords
T.
Aim: .
Consequences: .
Decisions: .
Abbreviations
T.
Aim: .
Consequences: .
Decisions: .
1. Introduction
The probe in the find, exploratory and development stages of drug research shows several constrictions which must be minimized in order to convey campaigner drugs faster into all-out production. Among these, dross profiling plays an of import function1. In fact, while pharmaceutical industry has been doing attempts to cut down the clip and cost that it takes to acquire merchandises to market, the potency for stableness and dross “surprises” that affect the development timeline has increased dramatically2.
From a chemical position, the being of pharmaceutical drosss are inevitable because no chemical reaction has 100 % selectivity – and no chemical compound is “rock” stable3. Some possible inauspicious effects that happen as a effect of this instability can be predicted and are given in Table 14.
Table 1: Potential Adverse Effects of Instability in Pharmaceutical Merchandises
|
Potential Adverse Consequence |
Explanation/Reason |
Example |
Stability Parameter Tested |
|
Loss of active ingredient |
Degradation of API in merchandise ensuing in less than 90 % drug as claimed on label – unacceptable quality |
Nitroglycerine tablets |
Time elapsed before the drug content no longer exceeds 90 % |
|
Addition in concentration of active ingredient |
Loss of vehicle perfusion bags sometimes allow dissolver to get away and vaporize so that the merchandise within the bag shows an addition in concentration |
Lidocaine gel, merchandises in perfusion bags |
Stability in concluding container |
|
Change in bioavailability |
Changes in rate extent of soaking up on storage |
— — — — — |
Dissolution/release surveies |
|
Loss of content uniformity |
Loss of contents as a map of clip |
Suspension |
Ease of re-dispersion or deposit volume |
|
Decline of microbiological position |
Addition in figure of feasible micro-organism already present in the merchandise. Contamination because of compromised bundle unity during distribution/storage |
Multiuse pick |
Entire bioburden after storage |
|
Loss of pharmaceutical elegance and patient acceptableness |
Stippling caused by interaction of the drug incorporating amine group with a minor constituent in the lactose resulting in the formation of a chromatophore |
Slight yellow or brown stippling on the surface of tablet incorporating spray-dried milk sugar |
Ocular Examination |
|
Formation of toxic debasement merchandises |
Degradation of the drug constituent |
Formation of epianhydrotetracycline from Achromycin, protein drugs |
Sum of debasement merchandises during shelf life |
|
Loss of bundle unity |
Change in bundle unity during storage or distribution |
Plastic screw cap losing back-off-torque |
Specific bundle unity trials |
|
Decrease of label quality |
Deterioration of label with clip and do the ink to run and therefore adversely affect discernability |
Plasticizer from fictile bottle migrates into the label |
Ocular scrutiny of the label |
|
Alteration of any factor of functional relevancy |
Time-dependent alteration of any functionally relevant property of a drug merchandise that adversely affects safety, efficaciousness, or patient acceptableness or easiness of usage |
Adhesion ripening of transdermic spots |
Monitoring alterations |
As easy understood, alterations in drug stableness can put on the line patient safety by formation of a toxic debasement merchandises or present a lower dosage than expected. Therefore, it is indispensable to cognize the pureness profile and behavior of a drug substance under assorted environmental conditions5.
This important information is gathered by proving the stableness of the drug merchandise, being portion of each stage of the drug development as it is required the clip period that the drug merchandise continues to keep its specification6. The development of a stableness bespeaking method ( SIM ) is indispensable to accurately mensurate the alterations in active ingredients concentration without intervention of other debasement merchandises, drosss and excipients7. In this field, forced debasement surveies play an of import function as they demonstrate the specificity of the developed method to mensurate the mentioned alterations when small information is available about possible debasement merchandise8.
Forced debasement surveies, emphasis testing, emphasis surveies, stress decomposition surveies, accelerated stableness or forced decomposition surveies are different ways to mention the same stableness trials mentioned2,9. Although these surveies have been in pattern in industry for a long clip, they merely became a formal regulative demand with the debut of the International Conference on Harmonization ( ICH ) guideline entitled “Stability Testing of New Drug Substances and Products” ( Q1A ) in 199310.
1.1. Forced debasement surveies
It is non easy to specify what a forced debasement survey is. It has become someway an “artful science” , as it is extremely dependent on the experience of the company and of the persons directing the surveies2. In add-on to this, many footings have been used interchangeably, like “forced degradation” and “accelerated stability”2.
In 1980, Pope defined accelerated stableness proving as “thevalidatedmethod or methods by which merchandise stableness may be predicted by storage of the merchandise under conditions, which accelerate alteration in a defined manner.”2. With the term “validated” , Pope meant that the alteration happening under the accelerated conditions must be demonstrated to correlate with normal long-run storage. More late, the International Conference for Harmonisation ( ICH ) introduced an of import differentiation between “forced degradation” and “accelerated stability” in the context of pharmaceutical stableness, indicating “accelerated testing” as11:
- Surveies designed to increase the rate of chemical debasement or physical alteration of an active drug substance or drug merchandise utilizing overdone storage conditions as portion of the formal, unequivocal, storage plan. These informations, in add-on to long-run stableness surveies, may besides be used to measure longer-term chemical effects at nonaccelerated conditions and to measure the impact of short-run jaunts outside the label storage conditions such as might happen during transporting. Consequences from accelerated proving surveies are non ever prognostic of physical alterations.
And “stress testing” as:
- Surveies undertaken to clarify the intrinsic stableness of the drug substance. Such testing is portion of the development scheme and is usually carried out under more terrible conditions than those used for accelerated testing.
It is clear that there is a distinction between both constructs, being stress proving distinguished by both badness of the conditions and the focal point or purpose of the consequences.
During a forced debasement trial, the undermentioned conditions should be investigated non merely in the drug substance but besides in the drug merchandise: acid and base hydrolysis ; hydrolysis at assorted pH ; thermic debasement ; photolysis ; and oxidization12.
Forced debasement surveies are designed to bring forth product-related discrepancies and develop analytical methods to find the debasement merchandises formed during accelerated pharmaceutical surveies and long-run stableness surveies. Any important debasement merchandise should be evaluated for possible jeopardy and the demand for word picture and quantification13,14.
There are many outcome that must be collected throw the forced debasement surveies15. Among them, we may stress:
- Appraisal of the stableness of the drug substance and drug merchandise ;
- Designation of structural transmutations of the drug substance and drug merchandise ;
- Detection of any low concentration of possible debasement merchandises ;
- Detection of unrelated drosss in the presence of the coveted merchandise and product-related degradants ;
- Separation of product-related degradants from those derived from excipients and integral placebo ;
- Elucidation about the possible debasement tracts ;
- Designation of debasement merchandises that may be spontaneously generated during the storage ;
- Facilitation of betterments in the fabrication procedure and preparations in analogue with accelerated pharmaceutical surveies
Although we can non albeit the importance of the stableness proving in the safety of the population, there is no standard process about how we should transport a forced debasement trial is available.
Questions that seem simple, like “How much stressing is adequate? ” are capable of much treatment amongst pharmaceutical scientists. Despite a value from 5 % to 20 % of debasement have been considered as sensible and acceptable for proof of chromatographic checks, some merchandises are so stable that small or no degradant is detected. Furthermore, if we consider expose the merchandise to energy in surplus of the energy provided by accelerated storage, it may take to overemphasizing and deviant consequences16–18.
1.1.1. Overview of regulative counsel
ICH have described forced debasement surveies in assorted international guidelines agreed between American, European and Nipponese regulative governments and followed by other regulative governments15. Among them, we may indicate:
- ICH Q1A ( R2 ) – Stability Testing of New Drug Substances and Merchandises11;
- ICH Q1B – Stability Testing: Photostability Testing of New Drug Substances and Merchandises19;
- ICH Q2 ( R1 ) – Validation of Analytical Procedures: Text and Methodology20;
The ICH Q1A ( R2 ) , in subdivision 2.1.2, reports the conditions for executing forced debasement surveies on drug substances and merchandises. This trial is likely to be carried out on a individual batch of the drug substance and include the consequence of temperature ( in 10°C increases ( e.g. , 50°C, 60°C, etc. ) above that for accelerated testing ) , humidness ( e.g. , 75 % comparative humidness or greater ) where appropriate, oxidization, and photolysis on the drug substance. The susceptibleness of the drug substance to hydrolysis across a broad scope of pH values should besides be evaluated when in solution or suspension. The scrutiny of the debasement merchandises under emphasis conditions is utile in set uping debasement tracts and developing and formalizing suited analytical processs. Although, if it is demonstrated that certain debasement merchandises are non formed under accelerated or long term storage conditions, there is no demand to analyze them.
The ICH Q1B contains recommendations for measuring the exposure stableness of drug substances and drug merchandises, being the conditions mentioned in Section 2 and Section 3, severally. No specific exposure degree are defined but it is stated that “samples should be exposed to light supplying an overall light of non less than 1.2 million lx hours and an integrated near ultraviolet energy of non less than 200 Ws hours/square metre to let direct comparings to be made between the drug substance and drug product.” . Despite the design of the survey is left for the applier, scientific justification must be given if the light exposure surveies are terminated after a short clip – may be related with the observation of inordinate debasement merchandise. Solid and solution/suspension may be subjected to the exposure stableness trials, being both used in the development of a stableness bespeaking method.
The ICH Q2 ( R1 ) gives counsel on how to formalize analytical methodological analysis and in subdivision 1.2.2 of Part II there is a recommendation to used samples from forced debasement surveies to turn out specificity when there are no drosss available.
Forced debasement surveies where non a regulative demand, although carried by the pharmaceutical industry a long clip ago, until the debut of the ICH guideline entitled “Stability Testing of New Drug Substances and Products” ( Q1A ) in 1993, the precursor of the referred version. Despite the Oklahoman the emphasis trials are held the easier is the choice of stability-indicating methods, guidelines does non explicitly necessitate its public presentation or study at the stage 1-2 investigational new drug ( IND ) phases7.The counsel require drug substance emphasis proving for Phase 3 IND but the regulative governments, nevertheless, ask inquiries refering the emphasizing consequences every bit early as a Phase 1 IND and suggest this surveies in drug merchandises2.
For a New Drug Application ( NDA ) , the counsel necessitate a sum-up of drug substance and drug merchandise emphasis surveies including elucidation of debasement tracts, presentation of the stability-indicating nature of analytical methods, and designation of important debasement merchandises21.It is besides required to emphasize the drug under hydrolytic, oxidative, photolytic and thermolytic conditions in solutions and in the solid province, as antecedently mentioned.
In the remainder of the universe, as there are many different regulative counsel, there are besides some considerations that should be stated9:
- Canada follow ICH guidelines with minor differences22;
- In South America, guidelines issued by Brazil and Mexico do non explicitly use the wordemphasis provingorforced decomposition, concentrating chiefly in long-run and accelerated storage. Although, debasement merchandises are emphasised23,2424;
- In Central America and Caribbean, Panama, El Salvador, Guatemala, Nicaragua, Costa Rica and Honduras have counsel similar to Mexico and Brazil25,26;
- In Africa, South Africa has adopted the ICH guidelines with minor alterations27. Republic of Kenya has a mix of ICH, EMA and WHO guidelines28. In Tanzania, one of their guideline clearly states that when we want to make some alterations in registered merchandises, certification should include “The stableness profile including the consequences on emphasis testing”29;
- In Golf Cooperation Council, which comprises Bahrain, Kuwait, Oman, Qatar, Saudi Arabia and the United Arab Emirates, the guidelines are parallel to WHO 2009 guideline30;
- Saudi Arabia define the demands really likewise to ICH QA1 ( R2 )31;
- The Association of Southeast Asiatic Nations, which includes Indonesia, Malaysia, Philippines, Singapore, Thailand, Brunei, Cambodia, Laos, Myanmar and Vietnam, describe the stress status as “40° C±2°C/75 % RH ± 5 % or at more nerve-racking conditions” ;
- In Singapore, emphasis testing is regulated as a necessity for assorted stairss of the drug development32;
- In India, the demands are really similar to the 1s stated by ICH33;
- Australia and New Zealand guidelines are besides similar to ICH Q1A ( R2 )9.
1.1.2. Review of bing literature and different attacks
As the drug debasement tracts differ from drug to drug, it is hard to make up one’s mind on the stressed conditions to be employed for a new drug at the clip of the induction of forced debasement surveies34. This can be understood if we think about the Arrhenius relationship – likely the most normally used look for measuring the relationship between rates of reaction and temperature for a given order reaction – as a manner to analyze the drug decomposition. If a drug obeys to the Arrhenius relationship, it is possible to gauge the consequence on temperature on the debasement rate of the compound, presuming that we know the energy of activation ( Tocopherola)35.
In 2011, MacFaul et Al. studied the dynamicss of debasement of 166 drug-like compounds in solution at elevated temperatures demoing that the mean Tocopherolawas 98.6 kJ/mol, within a scope from 49.8 kJ/mol to 197.5 kJ/mol36.
Datas from solid-state debasement surveies of more than 50 compounds in 100 surveies at Pfizer utilizing the Accelerated Stability Assessment Program ( ASAP ) attack indicated an mean Tocopherolaof 124.7 kJ/mol37.
This information demo how different drugs are and how hard it is to come up with a standard process for the stressed debasement surveies and different attacks have been used by pharmaceutical industry the last old ages38.
The design of these surveies must utilize more strenuous conditions than those used for accelerated surveies ( 25°C/60 % RH or 40°C/75 % RH ) . At a lower limit, acid and base hydrolysis, hydrolysis at assorted pH, thermic debasement, photolysis, and oxidization must be investigated21. Figure 1 can be used as a usher.

Figure 1: Flow diagram with the different forced debasement conditions to be used for drug substances and drug merchandises ( adapted from 16 )
Some writers have tried to stress how different are the conditions that drugs are subjected to during forced debasement surveies and the unsure reply we have for some important inquiries. S. Singh published in 2000 some counsel34seeking to reply the undermentioned inquiries:
- To what extreme of conditions one should travel if a antecedently tested status does non give sufficient debasement, good plenty for debasement merchandises to be isolated in measure suitable for word picture and construction elucidation?
- Are there any bounds where one should halt and transport no farther surveies?
- What kind of reagents or agents need to be employed for making a peculiar emphasis status?
As stated, each drug has a different attack to its forced debasement surveies and its debasement shows different rates. Although, the by and large recommended debasement varies between 5-20 % . This scope covers the by and large allowable 10 % debasement for little molecule pharmaceutical drug merchandises, for which the stableness bound is 90 % -110 % of the label claim5.
Although some scientists have found it practical to get down at utmost conditions ( 80°C or even higher, 0.5N NaOH, 0.5N HCl, 3 % H2Oxygen2)39, the conditions by and large employed for forced debasement surveies are the 1s presented in Table 2, published by George Ngwa16, because, as antecedently stated, overemphasizing can take to deviant consequences16–18.
Table 2: Conditionss Generally Employed for Forced Degradation
|
Degradation Type |
Experimental Condition |
Storage Condition |
Sampling Time |
|
Hydrolysis |
Control API ( no acid or base ) |
40°C, 60°C |
1, 3, 5 yearss |
|
0.1N HCL |
40°C, 60°C |
1, 3, 5 yearss |
|
|
0.1N NaOH |
40°C, 60°C |
1, 3, 5 yearss |
|
|
Acid Control ( no API ) |
40°C, 60°C |
1, 3, 5 yearss |
|
|
Base Control ( no API ) |
40°C, 60°C |
1, 3, 5 yearss |
|
|
pH: 2, 4, 6, 8 |
40°C, 60°C |
1, 3, 5 yearss |
|
|
Oxidative |
3 % H2Oxygen2 |
25°C, 40°C |
1, 3, 5 yearss |
|
Peroxide Control |
25°C, 40°C |
1, 3, 5 yearss |
|
|
Azobisisobutyronitrile ( AIBN ) |
40°C, 60°C |
1, 3, 5 yearss |
|
|
AIBN Control |
40°C, 60°C |
1, 3, 5 yearss |
|
|
Photolytic |
Light, 1 Ten ICH |
Sodium |
1, 3, 5 yearss |
|
Light, 3 Ten ICH |
Sodium |
1, 3, 5 yearss |
|
|
Light control |
Sodium |
1, 3, 5 yearss |
|
|
Thermal |
Heat Chamber |
60°C |
1, 3, 5 yearss |
|
Heat Chamber |
60°C/75 % RH |
1, 3, 5 yearss |
|
|
Heat Chamber |
80°C |
1, 3, 5 yearss |
|
|
Heat Chamber |
80°C/75 % RH |
1, 3, 5 yearss |
|
|
Heat Control |
Room Temperature |
1, 3, 5 yearss |
There are some other information that we should take into history refering the undermentioned subjects34:
- Drug concentration of the reaction solution
Some writers, harmonizing to their experience, recommend to utilize the concentration of 1 mg/mL. Although, in certain instances, the surveies must be carried at the concentration of the concluding preparation because the polymerisation dramas and of import function in the concluding concentration of the drug merchandise. As an illustration we have aminopenicillins and aminocephalosporins commercial readyings, where a scope of polymeric merchandises have been found to be formed when the drug is in high concentrations40.
- Managing of the reaction samples for the chromatographic surveies
Sometimes it is hard to cognize the best manner to manage samples that have a high concentration of acid, base or an oxidizing agent for shooting them into HPLC or lading on a TLC home base.
There are two chief attacks to cover with this state of affairs: dilution and neutralisation.
In the first one, it is advised that we should thin the sample plenty so that the concentration of reagent falls within the tolerable scope. In HPLC, the dilution can be done in the nomadic stage while utilizing TLC other dissolvers can be suited ( methyl alcohol, ethyl alcohol, etc. ) .
The 2nd one, involves the neutralisation of the acid and alkali solutions to a tolerable pH. Although, this has some drawbacks such as the trouble to transport the experiment out in a quantitative mode and the precipitation of the dissolved ingredients that sometimes occur.
As obvious, the recommended attack is the dilution, as it eliminates the drawbacks of the neutralisation. If there is any job with the dilution, it can be by and large solved by increasing the volume of injection or burden. This addition should be guided by the buffer capacity of the buffer used in the nomadic stage.
- The design of the surveies
For every emphasis survey, it is advised to bring forth four samples and describe the consequences of each: the clean solution stored under normal conditions, the space subjected to emphasize in the same mode as the drug solution, the nothing clip sample incorporating the drug which is stored under normal conditions and the drug solution subjected to emphasize intervention. A existent appraisal of alterations is merely made through the comparing of all these consequences.
- The equipment in usage
There are two of import equipment for emphasis testing: the container in which the reaction is done and the equipment for making the emphasis conditions. Each may change harmonizing to the reaction that is under analyzing ( hydrolytic, oxidative or photolytic ) .
For hydrolytic surveies in dilute acid and alkali conditions at temperatures between 5° C above room temperature up to 70° C, the reactions can merely be carried out in containers like volumetric flasks or stoppered civilization tubings and stored in a H2O bath set at the coveted temperature.
For reactions above 80° C and reflux conditions, one option is to utilize a boiling H2O bath equipped with a electromotive force regulator. Alternatively, an oil bath with a electromotive force regulator may be employed.
The oxidative emphasis surveies are suggested under normal research lab conditions merely. Hence no specific equipment is needed for the intent. The surveies should be done in a leak cogent evidence stoppered containers.
For photolytic reactions, the advice is to follow ICH guidelines. Hence any of the different sort of lamp beginnings defined in the guideline might be used. The end product of the lamp should run into D65/ID65 emanation criterions defined by ISO. The guideline suggests usage of unreal daylight fluorescent lamp uniting seeable and UV end products, Xe or metal halide lamp. Singh besides provided some utile information for scientists who are non certain how to carry on their forced debasement surveies: determination trees with the conditions to which the drug must be subjected in instance of hydrolysis, oxidization and photolysis ( Figure 4 and Figure 5 )34. The conditions for the survey of the consequence of temperature and humidness and photostability are already detailed in the ICH guidelines11,19.

Figure 2: Flow chart for executing emphasis surveies for hydrolytic debasement under acid and alkali conditions.

Figure 3: Flow chart for executing emphasis surveies for hydrolytic debasement under impersonal conditions.
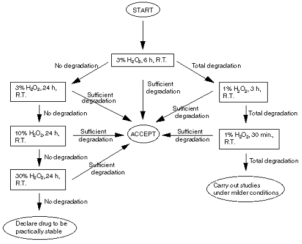
Figure 4: Flow chart for executing emphasis surveies for debasement under oxidative conditions.

Figure 5: Flow chart for executing emphasis surveies for photolytic debasement.
1.2. Furagin
1.2.1. Chemical and Physical Information
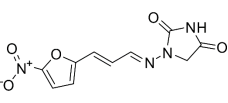
Figure 6: Furagin
CAS Registry Number: 0001672-88-441,42
IUPAC name: 1- [ ( E ) -3- ( 5-nitrofuran-2-yl ) prop-2-enylidene ] amino ] imidazolidine-2,4-dione
Synonym: Furazidine ; Furagin ; Solafur ; 1672-88-4 ; Akritoin ; NF 416 ;
Molecular Weight: 264.19432 g/mol
Molecular Formula: Degree centigrade10Hydrogen8Nitrogen4Oxygen5Boiling Point: 526.79°C
Hydrogen Bound Donor: 1Melting Point: 225.11°C
Hydrogen Bond Acceptor: 6Physical Form: Yellow Solid43
Solubility: soluble in DMSO43
1.2.2. Pharmaceutical Information
Furagin is a nitrofuran derivate, moving as an antibacterial medical specialty with bacteriostatic action. It is active against both Gram-positive (Staphylococcus epidermidis,Staphylococcus aureus,Staphylococcus faecalis) and Gram-negative micro-organisms (Enterobacteriaceae–Salmonella,Shygella,Proteus,Klebsiella,Escherichia,Enterobacter, etc. ) . It is used for intervention of ague and chronic urinary piece of land infections: pyelonephritis, cystitis, prostatitis, urethritis, and urinary postoperative infections: postoperative pyelonephritis, cystitis and prostatitis44.
1.2.3. Impurities
There are seven known drosss of Furagin45:
- Impurity A: 1- { ( E ) – [ ( 2E,4E ) -5- ( 5-nitrofuran-2-yl ) penta-2,4-dien-1-ylidene ] amino } -imidazolidine-2,4-dione ( Figure 7 )
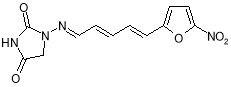
Figure 7: 1- { ( E ) – [ ( 2E,4E ) -5- ( 5-nitrofuran-2-yl ) penta-2,4-dien-1-ylidene ] amino } -imidazolidine-2,4-dione
Formed from 5- ( 5-nitrofuran-2-yl ) penta-2,4-dienal which is a known dross in ( E ) -3- ( 5-nitrofuran-2-yl ) propenal, an intermediate merchandise in the synthesis of Furagin. 5- ( 5-Nitrofuran-2-yl ) penta-2,4-dienal is formed from 5-nitrofurfurol by aldol crotonic condensation with ethanal through an intermediate ( E ) -3- ( 5-nitrofuran-2-yl ) propenal at the TP-2 phase of Furagin production. Then 5- ( 5-nitrofuran-2-yl ) penta-2,4-dienal reacts with 1-aminohydantoin hydrochloride ( 1?AHH ) at the TP-4 phase of Furagin production to give Impurity A ( Figure 8 )
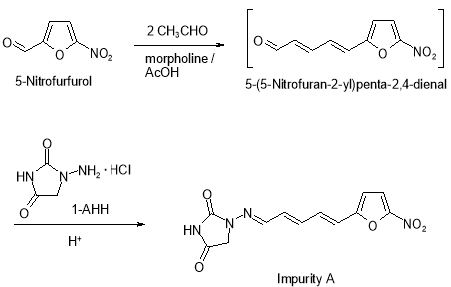
Figure 8: Chemical reaction strategy of dross A
- Impurity B: 1- [ (Omega) -3- ( 5-Nitro-furanyl-2 ) -propene-2- (Omega) -ylidenamino ] -imidazolidine-2,4-dione ( Figure 9 )
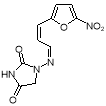
Figure 9: 1- [ ( Z ) -3- ( 5-Nitro-furanyl-2 ) -propene-2- ( Z ) -ylidenamino ] -imidazolidine-2,4-dione
Formed during the synthesis of Furagin by reaction of ( E ) -3- ( 5-nitrofuran-2-yl ) propenal, an intermediate merchandise of the synthesis of Furagin, with 1-aminohydantoin hydrochloride. Protonation of ( E ) -3- ( 5-nitrofuran-2-yl ) acrolein leads to formation of allylic cations in which a charge transportation between the terminals of the conjugate system and free rotary motion around the C2-C3 bond are possible. In this manner, E-double bond converts to Z- with the subsequent formation of the dross B after interaction with 1?aminohydantoin hydrochloride ( 1-AHH ) . So, this dross can be present in the Furagin API ( Figure 10 ) .
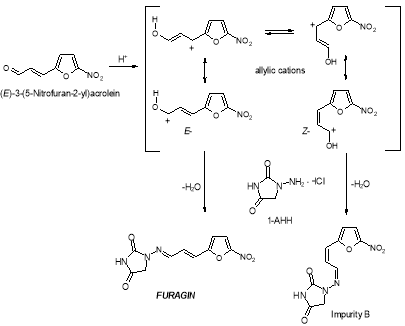
Figure 10: Chemical reaction strategy of dross B
Formation of Impurity B besides occurs upon irradiation of solutions of Furagin by visible radiation ( photoisomerization, photodegradation ) . Reverse procedure of formation of Furagin from Impurity B is besides possible under irradiation ( Figure 11 ) .

Figure 11: Chemical reaction strategy of the formation of Furagin from Impurity B
- Impurity C: 5-Nitrofuran-2-carbaldehyde ( Figure 12 )
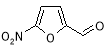
Figure 12: 5-Nitrofuran-2-carbaldehyde
5-Nitrofurfural is a man-made intermediate which can be carried over to Furagin API. 5?Nitrofurfural is formed by acerb catalyzed hydrolysis of 5-nitrofurfural diacetate ( 5-NFDA ) at the TP-1 phase of Furagin production ( Figure 13 ) .
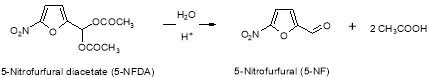
Figure 13: Chemical reaction strategy of dross C
- Impurity D: 3- ( 5-Nitrofuran-2-yl ) prop-2-enal ( Figure 14 )
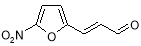
Figure 14: 3- ( 5-Nitrofuran-2-yl ) prop-2-enal
?- ( 5-Nitro-2-furyl ) propenal is an intermediate in the synthesis of Furagin and can be carried over to Furagin API. The strategy of formation of ?- ( 5-nitro-2-furyl ) propenal at the phase TP-2 of Furagin production is provided below ( Figure 15 ) .
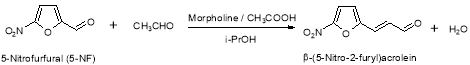
Figure 15: Chemical reaction strategy of dross D
?- ( 5-Nitro-2-furyl ) propenal may besides be formed as degradant in an acid-catalyzed hydrolysis of Furagin ( Figure 16 ) .
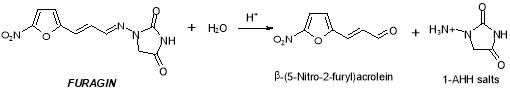
Figure 16: Chemical reaction strategy of the formation of dross D by acid catalysed hydrolysis of Furagin
- Impurity E: 1- { ( E ) – [ ( 5-nitrofuran-2-yl ) methylidene ] amino } -imidazolidine-2,4-dione ( Figure 17 )
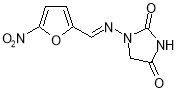
Figure 17: 1- { ( E ) – [ ( 5-nitrofuran-2-yl ) methylidene ] amino } -imidazolidine-2,4-dione
This dross may be formed at the TP-4 phase of Furagin production by reaction of 5-nitrofurfural ( possible dross in ?- ( 5-nitro-2-furyl ) propenal ) with 1-aminohydantoin hydrochloride ( 1-AHH ) ( Figure 18 ) .
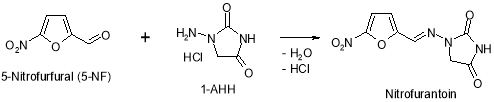
Figure 18: Chemical reaction strategy of dross E
- Impurity F: { ( 2E ) -1-carbamoyl-2- [ ( 2E ) -3- ( 5-nitrofuran-2-yl ) prop-2-en-1-ylidene ] hydrazinyl } -acetic acid ( Figure 19 )
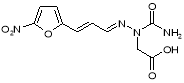
Figure 19: { ( 2E ) -1-carbamoyl-2- [ ( 2E ) -3- ( 5-nitrofuran-2-yl ) prop-2-en-1-ylidene ] hydrazinyl } -acetic acid
This dross may be formed at the TP-4 phase of Furagin production by reaction of 2-semicarbazide-acetic acid ( possible dross in 1-aminohydantoin hydrochloride ) with ?- ( 5-nitro-2-furyl ) propenal ( Figure 20 ) .
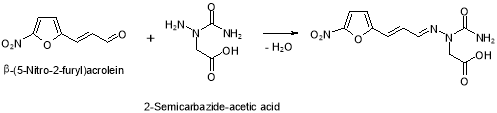
Figure 20: Chemical reaction strategy of dross F
This dross can besides be formed as degradant by base-catalyzed hydrolysis of Furagin ( Figure 21 ) .
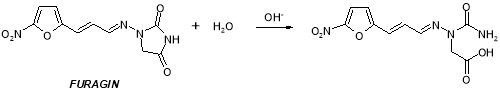
Figure 21: Chemical reaction strategy of the formation of dross F by basic catalysed hydrolysis of Furagin
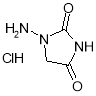
- Impurity G: 1-Aminoimidazolidine-2,4-dione hydrochloride ( Figure 22 )
Essay Writing Service Fully referenced, delivered on clip, Essay Writing Service. Assignment Writing Service Everything we do is focussed on composing the best possible assignment for your exact demands Taging Service Our Marking Service will assist you pick out the countries of your work that need betterment. View our services FREE APA Referencing Tool Create your 6th Edition APA mentions rapidly, easy and for free! FREE Harvard Referencing Tool Our free online Harvard Referencing Tool makes citing easy. Free Vancouver Referencing Tool Get aid with your Vancouver referencing with our free online tool. FREE Study Guides Everything you need to cognize during your surveies Place an order now Our experts are waiting to assist you with your essay Our experts can assist you with your essay question Start your order No thanks
Furagin: Forced Degradation Studies. (2017, Aug 11). Retrieved from https://paperap.com/paper-on-furagin-forced-degradation-studies/
